How to Cite | Publication History | PlumX Article Matrix
Nabomita Paul1 , Ajay Pandey2
, Ajay Pandey2 and Khushhali M. Pandey3
and Khushhali M. Pandey3
1Department of Biotechnology, Sri Sathya Sai College for Women, Bhopal, 462024, India.
2Department of Mechanical Engineering, MANIT, Bhopal, 462003, India.
3Department of Biological Science and Engineering, MANIT, Bhopal, 462003, India.
Corresponding Author E-mail: menaria.khushhali@gmail.com
DOI : http://dx.doi.org/10.13005/bbra/2737
ABSTRACT: Diabetes is a consequential human ailment afflicting a significant population in many countries across the world. In India, it is turning out to be a major health quandary, especially in urban areas. Though there are various approaches to reduce the ill-effects of diabetes and its secondary complications, herbal formulations are preferred due to lesser side-effects and low cost. Traditional medicines derived from medicinal plants are utilized by about 60% of the world’s population. This review focuses on Indian herbal drugs and medicinal plants utilized in the treatment of diabetes, especially in India.
KEYWORDS: Antidiabetic; Antioxidant; Diabetes; Medicinal Plants; Sulfonylurea; Thiazolidinediones
Download this article as:| Copy the following to cite this article: Paul N, Pandey A, Pandey K. M. An Understanding of Diabetes Mellitus Associated Complications, Treatment Modalities and Management Strategies. Biosci Biotech Res Asia 2019;16(1). |
| Copy the following to cite this URL: Paul N, Pandey A, Pandey K. M. An Understanding of Diabetes Mellitus Associated Complications, Treatment Modalities and Management Strategies. Biosci Biotech Res Asia 2019;16(1). Available from: https://bit.ly/2Ua7dhc |
Introduction
Diabetes is a chronic disorder in the metabolism of proteins, fats, and carbohydrates.1,2 It is described as an increase in blood glucose levels after consumption of any type of meal. Diabetes results from either insulin deficiency or its malfunctioning.2 According to statistics, 2.8% of the world’s population suffers from this disease and it is expected to increase to more than 5.4% by 2025.3 Diabetes requires early diagnosis, treatment, and lifestyle changes. Diabetes is a disease that affects many people in the 21st century and is known as the ‘fifth’ leading cause of death.4 High prevalence, variable pathogenesis, progressive process, and complications of diabetes all highlight the urgent need for effective treatments. Nowadays, different treatments, such as insulin therapy, pharmacotherapy, and diet therapy, are available to control diabetes. There are several types of glucose-lowering drugs that exert anti-diabetic effects through different mechanisms. These mechanisms include stimulation of insulin secretion by sulfonylurea and meglitinides drugs, increasing of peripheral absorption of glucose by biguanides and thiazolidinediones,5 delay in the absorption of carbohydrates from the intestine by alpha-glucosidase, and reduction of hepatic gluconeogenesis by biguanides.6
Table 1: History of Diabetes mellitus.
| Year | Event |
| 1889 | The discovery of the role of the pancreas in diabetes was made by Joseph Von Mering and Oskar Minkowski. They found that upon complete removal of the pancreas from dogs, the dogs exhibited all the signs and symptoms of diabetes and died shortly afterwards [7]. |
| 1910 | Sir Edward Albert Sharpey-Schafer of Edinburgh in Scotland suggested that diabetics lacked a single chemical which was normally produced by the pancreas. Name of this chemical was later proposed to be insulin [7]. |
| 1921 | Frederick Grant Banting and Charles Herbert Best repeated the work of Von Mering and Minkowski but went a step further and managed to show that they could reverse the induced diabetes in dogs by giving them an extract from the pancreatic islets of Langerhans of healthy dogs. This was a step forward in elucidation of the endocrine role of the pancreas in metabolism and existence of insulin [8]. |
| 1922 | Scientists proceeded on to isolate insulin from bovine pancreases at the University of Toronto in Canada, thereby leading to the availability of an effective treatment of diabetes mellitus, with the first clinical patient being treated [7]. |
| 1935 | The distinction between what is now known as type I and type II diabetes was made by Sir Harold Percival (Harry) Himsworth [7]. |
Molecular Mechanism of Insulin Signalling
Insulin is an anabolic peptide hormone released by the β-pancreatic islet cells in response to elevated levels of nutrients, such as glucose in the blood supply. Insulin binds to its receptor which belongs to the receptor tyrosine kinase superfamily. It has orthologues in all metazoans on the major insulin-responsive tissues of the body namely skeletal muscle, adipose tissue (where it stimulates glucose transport through translocation of GLUT4), liver (where it promotes glucose storage into glycogen and decreases glucose output), and in most cells where it has pleiotropic effects. The receptor activates a complex intracellular signalling network through Insulin receptor substrates (IRS) termed phosphotyrosine-binding (PTB) domain of adaptor proteins and the canonical PI3K and ERK cascades whose function is to stimulate the transport of nutrients (glucose, amino acids and fatty acids) from the blood supply to tissues and then to promote the conversion of nutrients into storage macromolecules (glycogen, protein, and lipids). Insulin receptor resists a hetero-tetrameric structure (two extracellular α- subunits and two transmembrane β-subunits). When insulin binds to the α-subunits, it activates the β- subunits & these β-subunits auto-phosphorylate themselves on tyrosine residues. Basically, once the β-subunits are phosphorylated they get activated and lead to the phosphorylation and activation of insulin receptor substrate (IRS1/2). IRS1/2 activation and dephosphorylation is regulated by a protein called p10 or phosphatase intense and homologue effaced from chromosome 10. This is a negative regulator of pip3 that results in dephosphorylation of pip3. The PI3K-AKT pathway, mostly responsible for the metabolic insulin action via the translocation of the glucose transporter type 4 (GLUT4) vesicles to the plasma membrane, which, in turn, allows the glucose uptake in muscle cells and adipocytes. PI3K or tri phosphatidylinositol 3 kinase protein binds to IRS1/2 through its p85 subunit, the PI3K protein will phosphorylate pip2 or Phosphatidylinositol 4,5-bisphosphate to pip3 or Phosphatidylinositol (3,4,5)-trisphosphate. When pip3 concentration increases, it recruits & activates other proteins (PDK1 & AKT or protein kinase B) towards the plasma membrane. After activation of PDK1, it will phosphorylate AKT. When insulin concentrations are low, insulin sensitive cells have reservoirs of vessels (intracellular vesicles) that contain glucose conveyors embedded in the cytoplasmic vesicles (GLUT 4), which moves towards the plasma membrane with the avails of the protein called AS160. GLUT4 – glucose transporters are present in a vesicle, but they are useless for transporting glucose. After inhibition of glucose through vascular translocation & insulin stimulation, AKT will phosphorylate and inactivate AS160 which will allow translocation to occur. Glucose transporter will get embedded into a cellular membrane after vascular translocation. AKT phosphorylates other proteins like GSK3 or glycogen synthase kinase 3 & it will allow inhibition of GSK3 which is a kinase inactivates GS or glycogen synthase (an important enzyme involved in glycogen synthesis) after GSK3 phosphorylates GS. The TSC1/2-mTOR pathway, playing a critical role in protein synthesis since the mammalian target of rapamycin (mTOR) is a central controller for several anabolic and catabolic processes including RNA translation, ribosome biogenesis and autophagy, in response not only to growth factors and hormones like insulin but also to nutrients, energy and stress signals.GLUT4 allows glucose to get into the cell where glucose can undergo glycolysis. AKT not only allow glucose to enter the cell & undergo glycolysis but it’s also activating glycogen synthesis (to store glucose which is in the cell). AKT activates mTORC1 or mammalian target of rapamycin complex 1, including steps like tuber sclerosis complex, reb proteins, etc. After activation of mTORC1, it will activate its downstream target – P70S65 kinase through phosphorylation. P70S55 negatively inhibit IRS proteins by phosphorylating IRS on serine residues. This is how the insulin signalling is controlled by negative feedback inhibition. GLUT4 localisation in the plasma membrane is entirely dependent on the presence of insulin and activation of insulin receptor so when insulin is dislodged from the insulin receptor, GLUT4 is taken back up into vesicles which contain them for the next time the cells stimulated by insulin. There is some tissue that does not require insulin for efficient uptake of glucose: for instance – the brain and the liver. This is because these cells don’t use GLUT4 for importing glucose, but use another transporter that is not insulin-dependent. The insulin signalling pathway (ISP) is an important biochemical pathway, which regulates some fundamental biological functions (glucose and lipid metabolism, protein synthesis, cell proliferation, cell differentiation and apoptosis).
Molecular Mechanism and Genetics Affected in Diabetes Mellitus
Diabetes is controlled by lowering the hepatic glucose production. It is a primary therapeutic strategy to control diabetes. Increased hepatic glucose production in diabetic states may occur as a consequence of insulin deficiency or insulin resistance that impairs glucose uptake in multiple organs. Most of the strategies employed to reduce hepatic glucose production in diabetes are associated with stimulation of the energy sensor enzyme AMPK (adenosine monophosphate-activated protein kinase), which is normally activated by an elevated AMP to ATP ratio to block cellular anabolism (i.e., gluconeogenesis, lipogenesis, glycogen synthesis). AMPK is taken into account as a master switch regulating glucose and macromolecule metabolism. The AMPK is an enzyme that works as a fuel gauge, being activated in conditions of high energy phosphate depletion. AMPK is additionally activated robustly by muscle contraction and heart muscle anaemia and is concerned within the stimulation of glucose transport and carboxylic acid-reaction made by these stimuli. In the liver, activation of AMPK ends up in increased fatty acid reaction and reduced production of glucose, cholesterol, and triglycerides. The two leading diabetic medications, namely – metformin and rosiglitazone, show their metabolic effects partly through AMPK. The amp-stimulated enzyme, AMPD2, is a natural counter-regulator of AMPK and its activation in the liver of diabetic mice is associated with reduced AMPK activity.
Diabetes Type-1 risk is increased by certain variants of the HLA-DQA1, HLA-DQB1, and HLA-DRB1 genes. These genes provide instructions for making proteins that play a critical role in the immune system. They belong to a family of genes called the Human Leukocyte Antigen (HLA) complex. The HLA complex helps the immune system distinguish the body’s own proteins from proteins made by foreign invaders such as viruses and bacteria.
Diabetes Type-2 arises when pancreatic β-cells fail to secrete sufficient insulin, because of acquired β-cell secretory dysfunction or decreased β-cell mass. β-cell mass plays a vital role in determining whether an individual will progress to diabetes. These defects may be caused by primary β-cell defects, such as the one seen in the monogenic diabetes forms of MODY (Maturity-Onset Diabetes of the Young), or by secondary β-cell defects, caused by glucotoxicity, increased free fatty acids, cytokines, mitochondrial dysfunction or metabolic stress.
Genetic factors – After the explanation of Mendelian disorders with diabetes as a major phenotypic feature, it has become clear that type-2 diabetes is heterogeneous and may result from defects in one or more molecular pathways. Genetic defects of the β-cell usually referred to as Maturity-Onset Diabetes of the Young (MODY), can result from mutations in any of at least six different genes. Most of the MODY subtypes are caused by mutations in transcription factors, which are involved in the tissue-specific regulation of gene expression in the liver and in pancreatic beta-cells. Other related genetic factors are due to insulin receptor mutations. Gene variants may impair insulin action in the insulin target tissues muscle, fat and liver.
Table 2: Type 2 diabetes mellitus susceptibility genes.
| Gene | Function | References |
| ADAMTS9 | Metalloproteinase/Insulin action | [9–11] |
| ADCY5 | Adenylyl cyclases/Insulin action | [12] |
| ANK1 | Cell stability/β-cell function | [13–15] |
| ANKRD55 | Insulin action | [13,14] |
| ANKS1A | Pathway regulator/unknown | [16] |
| BCAR1 | Docking protein/Beta cell function | [13,14] |
| BCL2 | Cell death regulator/unknown | [11] |
| BCL11A | Zinc finger/β-cell function | [9] |
| CAMK1D | Protein kinase/β-cell function | [9-11] |
| CAPN10 | Calpain cysteine protease/insulin action | [17–20] |
| CENTD2 | β-cell function | [9,11] |
| CDKAL1 | β-cell function | [11,21-23] |
| CDKN2A
|
Cyclin-dependent kinase inhibitor
/ Beta-cell function |
[11,21,22] |
| DGKB | Diacylglycerol kinase/insulin action | [11,12] |
| DUSP9 | Phosphatase | [9,11] |
| FOLH1 | Transmembrane glycoprotein/unknown | [11] |
| FTO | Metabolic regulator/Insulin action | [11,24] |
| GCK | Glucokinase/Insulin action | [12] |
| GCKR | Glucokinase regulator/Insulin action | [11,12] |
| GIPR | G-protein coupled receptor | [11] |
| SLC30A8 | Zinc efflux transporter/beta cell function | [9-12,21,29] |
| GRB14 | Adapter protein/Insulin action | [13,14] |
| HFE | Membrane protein | [25] |
| HHEX | Transcriptional repressor | [9,11,21,26] |
| HNF1α | Pancreatic and liver transcriptional activator | [9,11] |
| IRS 1 | Insulin signalling element/Insulin action | [9,11,27] |
| PPARG | Nuclear receptor/Insulin action | [9,11,21,28] |
| PRC1 | Cytokinesis regulator | [9] |
Management of Diabetes Mellitus
Lifestyle management is apparently the cornerstone of management of diabetes mellitus. It is recognized as the essential part of diabetes prevention. Meta-analyses demonstrate that manner interventions, as well as diet and physical activity, led to a 63% reduction in polygenic disorder incidence in those at high risk. Lifestyle modification programs have incontestable encouraging improvement in risk factors for diabetes; but, the impact on diabetes incidence has not been reportable. The dietary management of diabetes could be a complement of lifestyle management. It’s a positive impact on future health and quality of life. Dietary management aims at best metabolic management by establishing a balance between food intake, physical activity, and drugs to avoid complications. In type-2 diabetes, the dietary objective is for improved glycemic and macromolecule levels and weight loss as acceptable. Plant-derived medications have additionally found vast use within the management of diabetes.
![Figure 1: Types of Diabetes Mellitus [48-58].](https://www.biotech-asia.org/wp-content/uploads/2019/04/Vol_16_no1_Und_Nab_fig1-150x150.jpg) |
Figure 1: Types of Diabetes Mellitus [48-58].
|
![Figure 2: Diabetes Prevalence [59].](https://www.biotech-asia.org/wp-content/uploads/2019/04/Vol_16_no1_Und_Nab_fig2-150x150.jpg) |
Figure 2: Diabetes Prevalence [59].
|
Diabetic Complications
Acute Complications
These types of complications include diabetic ketoacidoses (DKA) and non-ketotic hyperosmolar state (NKHS). While the first is seen primarily in individuals with type-1 diabetes mellitus, the latter is prevalent in individuals with type-2 diabetes mellitus. In DKA, sickness and retching are frequently present. Torpidity and focal sensory system gloom may develop into a trance-like state in extreme DKA. Cerebral oedema, a genuine complication, is usually seen in youngsters. NKHS is generally found in older people. NKHS is most commonly seen in elderly individuals with type-2 diabetes mellitus.
![Figure 3: History of Diabetes Medications [60-68].](https://www.biotech-asia.org/wp-content/uploads/2019/04/Vol_16_no1_Und_Nab_fig3-150x150.jpg) |
Figure 3: History of Diabetes Medications [60-68].
|
Chronic Complications
The chronic complications of diabetes mellitus affect many organ systems and are responsible for the majority of morbidity and mortality. Chronic complications can be divided into vascular and nonvascular complications.
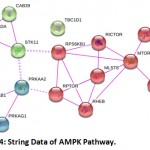 |
Figure 4: String Data of AMPK Pathway.
|
Therapeutics of Diabetes Mellitus
Caloric Content
Most patients with non-insulin dependent diabetes mellitus (NIDDM) are overweight or obese, and it is now well-recognized that this is a major factor in insulin resistance. When extreme caloric restriction and/or rapid weight loss seem desirable, a very low-calorie diet or protein-sparing modified fast may be considered. Reduction of excess weight is a primary component in the management of NIDDM.
![Figure 5: Diabetic Complications and its Diseases [69-73].](https://www.biotech-asia.org/wp-content/uploads/2019/04/Vol_16_no1_Und_Nab_fig5-150x150.jpg) |
Figure 5: Diabetic Complications and its Diseases [69-73].
|
Macronutrients
The ideal balance of carbohydrate, protein, or fat intake in patients with NIDDM is still a matter of discussion. It has recently been recognized that a diet containing 60% carbohydrates, even if not including sugar, may predispose to the development of dyslipidemia. Protein intake should not exceed the daily requirement since high protein intake appears to have a detrimental effect on renal function.
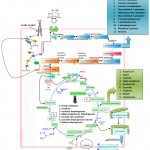 |
Figure 6: Molecular Mechanism of Insulin Signaling.
|
Physical Activity and Exercise
Recent clinical investigations have shed light on the mechanism by which exercise may help in controlling excessive blood glucose levels. Regular exercise improves insulin sensitivity and, as a consequence, may improve glucose tolerance. Such effects are beneficial in patients with type-2 diabetes since they enhance work capacity and quality of life and may also help to reduce the requirement for insulin or oral hypoglycemic agents.
Table 3: Herbal Drug Formulations[30]
| Drug | Company | Ingredients |
| Diabecon | Himalaya | Abutilon indicum, |
| Aloe vera, | ||
| Asparagus racemosus, | ||
| Berberis aristata, | ||
| Boerhavia diffusa, | ||
| Casearia esculenta, | ||
| Commiphora wightii, | ||
| Curcuma longa, | ||
| Glycyrrhiza glabra, | ||
| Gmelina arborea, | ||
| Gossypium herbaceum, | ||
| Gymnema sylvestre, | ||
| Momordica charantia, | ||
| Ocimum sanctum, | ||
| Phyllanthus amarus, | ||
| Piper nigrum, | ||
| Pterocarpus marsupium, | ||
| Rumex maritimus, | ||
| Shilajeet, | ||
| Sphaeranthus indicus, | ||
| Swertia chirata, | ||
| Syzygium cumini, | ||
| Tinospora cordifolia, | ||
| Tribulus terrestris, | ||
| Triphala. | ||
| Ayurveda alternative | Chakrapani Ayurveda | Azadirachta indica, |
| Chakrapani Ayurved | Gymnema sylvestre, | |
| herbal formula to | Inula racemosa, | |
| Diabetes: | Momordica charantia, | |
| Bitter gourd Powder | Garry and Sun natural | Momordica charantia, |
| Remedies | Syzygium cumini, | |
| Tinospora cordifolia, | ||
| Trigonella foenum gracecum. | ||
| Diabetes-Daily Care | Nature’s Health Supply | Alpha Lipoic Acid, |
| Chromax, | ||
| Cinnamon 4% Extract, | ||
| Fenugreek 50% extract, | ||
| Gymnema sylvestre 25% extract, | ||
| Licorice Root 20% extract, | ||
| Momordica 7% extract, | ||
| Vanadium. | ||
| Gurmar powder | Garry and Sun natural | Gymnema Sylvestre. |
| Remedies |
Table 4: Medicinal plants with antidiabetic & related beneficial properties.
| Plant name & Family | Activity | Reference |
| Andrographis paniculata(Burm.f.)Wall Acanthaceae | It has shown antidiabetic and antihyperlipidemic effects. Its active compound is andrographolide & it shows hypoglycemic and hypolipidemic effects. | [31] |
| Ageratum conyzoides Asteraceae | It has hypoglycemic effects. | [32] |
| Swertia chirata Gentianaceae | It has a beneficial effect on cholesterol and triglyceride level. | [33] |
| Terminalia arjuna Combretaceae | It decreases the activity of glucose-6-phosphatase, fructose-1,6-bisphosphatase, aldolase and an increase in the activity of phosphoglucoisomerase and hexokinase in tissues. | [34] |
| Azadirachta indica Meliaceae | It has anti-diabetic activity, anti-bacterial, antimalarial, antifertility, hepatoprotective and antioxidant effects. | [35] |
| Alangium lamarckii
Alangiaceae |
It has significant antidiabetic activity in nicotinamide induced diabetic rat. | [36] |
| Albizia odoratissima Mimosaceae
|
Significantly reduces the levels of serum cholesterol, triglycerides, alkaline phosphatase and decrement of total proteins in alloxan induced albino mice. | [37] |
| Chaenomeles Sinensis Rosaceae | Ethyl acetate fraction shown promising antidiabetic effect. | [38] |
| Acacia arabica Leguminosae | It induces hypoglycemia. | [39] |
| Aegle marmelos
Rutaceae |
IT possesses hypoglycemic activity and prevents the peak rise in blood sugar. | [40] |
| Allium cepa Amaryllidaceae | It has significant antioxidant and hypolipidaemic activity. | [41,42,43] |
| Aloe barbadensis Liliaceae | It effectively increases glucose tolerance. | [44-45] |
| Stevia rebaudiana Asteraceae | Steviol glycosides stimulate the insulin secretion through potentiation of the β-cell, preventing high blood glucose after a meal. | [46-47] |
Table 4: Medicinal plants with antidiabetic & related beneficial properties.
| Plant name & Family | Activity | Reference |
| Andrographis paniculata(Burm.f.)Wall Acanthaceae | It has shown antidiabetic and antihyperlipidemic effects. Its active compound is andrographolide & it shows hypoglycemic and hypolipidemic effects. | [31] |
| Ageratum conyzoides Asteraceae | It has hypoglycemic effects. | [32] |
| Swertia chirata Gentianaceae | It has a beneficial effect on cholesterol and triglyceride level. | [33] |
| Terminalia arjuna Combretaceae | It decreases the activity of glucose-6-phosphatase, fructose-1,6-bisphosphatase, aldolase and an increase in the activity of phosphoglucoisomerase and hexokinase in tissues. | [34] |
| Azadirachta indica Meliaceae | It has anti-diabetic activity, anti-bacterial, antimalarial, antifertility, hepatoprotective and antioxidant effects. | [35] |
| Alangium lamarckii
Alangiaceae |
It has significant antidiabetic activity in nicotinamide induced diabetic rat. | [36] |
| Albizia odoratissima Mimosaceae
|
Significantly reduces the levels of serum cholesterol, triglycerides, alkaline phosphatase and decrement of total proteins in alloxan induced albino mice. | [37] |
| Chaenomeles Sinensis Rosaceae | Ethyl acetate fraction shown promising antidiabetic effect. | [38] |
| Acacia arabica Leguminosae | It induces hypoglycemia. | [39] |
| Aegle marmelos
Rutaceae |
IT possesses hypoglycemic activity and prevents the peak rise in blood sugar. | [40] |
| Allium cepa Amaryllidaceae | It has significant antioxidant and hypolipidaemic activity. | [41,42,43] |
| Aloe barbadensis Liliaceae | It effectively increases glucose tolerance. | [44-45] |
| Stevia rebaudiana Asteraceae | Steviol glycosides stimulate the insulin secretion through potentiation of the β-cell, preventing high blood glucose after a meal. | [46-47] |
Conclusion
Plant drugs and herbal formulations are considered to be less toxic and free from side-effects than synthetic ones. Based on the WHO recommendations, hypoglycaemic agents of plant origin used in medicine are important. By 2025, it is predicted the majority of diabetes – positive people of developed countries will be age of 45 – 64 and will be affected in their most productive years in developing countries. In general, there is very little biological erudition on the categorical modes of action in the treatment of diabetes, but most of the plants have been found to contain substances like glycosides, alkaloids, terpenoids, flavonoids etc. that are frequently implicated as having anti-diabetic effects. The research for alternate remedies from the plant kingdom for diabetes mellitus will continue all over the world as the dreaded disease poses many challenges not only to the physician but also to the researcher.
References
- Osadebe P. O., Odoh E. U., Uzor P. F. The search for new hypoglycemic agents from the plant. Afr. J. Pharm. Pharmacol. 2014;8(11):292-303.
CrossRef - Modak M., Dixit P., Londhe J., Ghaskadbi S., Devasagayam T. P. A. Indian herbs and herbal drugs used for the treatment of diabetes. J. Clin. Biochem. Nutr. 2007;40(3):163.
CrossRef - Mukesh R., Namita P. Medicinal Plants with Antidiabetic Potential-A Review. American-Eurasian J. Agric. Environ. Sci. 2013; 13(1):81-94.
- Kazi S. Use of traditional plants in diabetes mellitus. Int. J. Pharm. 2014;4(4):283-9.
- Bathaie S., Mokarizade N., Shirali S. An overview of the mechanisms of plant ingredients in the treatment of diabetes mellitus. J. Med. Plant. 2012;4(44):1-24.
- Hui H., Zhao X., Perfetti R. Structure and function studies of glucagon-like peptide-1 (GLP1): the designing of a novel pharmacological agent for the treatment of diabetes. Diabetes Metab. Res. Rev. 2005;21;313-31.
CrossRef - Himsworth H. P. Diabetes mellitus: its differentiation into insulin-sensitive and insulin-insensitive types. Lancet. 1936; 227(5864);127-130.
CrossRef - Banting F. G., Best C. H., Collip J. B., Campbell W. R and Fletcher A. A. Pancreatic extracts in the treatment of diabetes mellitus-Preliminary report. Canadian Med. Association Journal. 1922;145(10);1281-1286.
- Voight B. F., Scott L. J., Steinthorsdottir V., Morris A. P., Dina C.,Welch R. P., Zeggini E., Huth C., Aulchenko Y. S., Thorleifsson G., McCulloch L. J., Ferreira T., Grallert H., Amin N., Wu G., Willer C. J., Raychaudhuri S., McCarroll S. A., Langenberg C.,Hofmann O. M., Dupuis J., Qi L., Segrè A. V., van Hoek M., Navarro P., Ardlie K., Balkau B., Benediktsson R.,Bennett A. J., Blagieva R., Boerwinkle E., Bonnycastle L. L., Bengtsson B. K., Bravenboer B., Bumpstead S., Burtt N. P., Charpentier G., Chines P. S., Cornelis M., Couper D. J., Crawford G., Doney A. S.,Elliott K. S., Elliott A. L., Erdos M. R., Fox C. S., Franklin C. S., Ganser M., Gieger C., Grarup N., Green T., Griffin S., Groves C. J., Guiducci C., Hadjadj S., Hassanali N., Herder C., Isomaa B., Jackson A. U., Johnson P. R., Jørgensen T., Kao W. H., Klopp N., Kong A., Kraft P., Kuusisto J., Lauritzen T., Li M., Lieverse A., Lindgren C. M., Lyssenko V., Marre M., Meitinger T., Midthjell K., Morken M. A., Narisu N., Nilsson P., Owen K. R., Payne F., Perry J. R., Petersen A. K., Platou C., Proença C., Prokopenko I., Rathmann W., Rayner N. W., Robertson N. R., Rocheleau G., Roden M., Sampson M. J., Saxena R., Shields B. M., Shrader P., Sigurdsson G., Sparsø T., Strassburger K., Stringham H. M., Sun Q., Swift A. J., Thorand B., Tichet J., Tuomi T., VanDam R. M., Van Haeften T. W., Van H. T., Van Vliet-Ostaptchouk V., Walters G. B., Weedon M. N., Wijmenga C., Witteman J., Bergman R. N., Cauchi S., Collins F. S., Gloyn A. L., Gyllensten U., Hansen T., Hide W. A., Hitman G. A., Hofman A., Hunter D. J., Hveem K., Laakso M., Mohlke K. L., Morris A. D., Palmer C. N., Pramstaller P. P., Rudan I., Sijbrands E., Stein L. D., Tuomilehto J., Uitterlinden A., Walker M., Wareham N. J., Watanabe R. M., Abecasis G. R., Boehm B. O., Campbell H., Daly M. J., Hattersley A. T., Hu F. B., Meigs J. B., Pankow J. S., Pedersen O., Wichmann H. E., Barroso I., Florez J. C., Frayling T. M., Groop L., Sladek R., Thorsteinsdottir U., Wilson J. F., Illig T., Froguel P., van Duijn C. M., Stefansson K., Altshuler D., Boehnke M., McCarthy M. I., Twelve type 2 diabetes susceptibility loci identified through large-scale association analysis. Nat. Genet. 2010;42:579–589.
CrossRef - Zeggini E., Scott L. J., Saxena R., Voight B. F., Marchini J. L., Hu T., de Bakker P. I., Abecasis G. R., Almgren P., Andersen G., Ardlie K., Boström K. B., Bergman R. N., Bonnycastle L. L., Borch J. K., Burtt N. P., Chen H., Chines P. S., Daly M. J., Deodhar P., Ding C. J., Doney A. S., Duren W. L., Elliott K. S., Erdos M. R., Frayling T. M., Freathy R. M., Gianniny L., Grallert H., Grarup N., Groves C. J., Guiducci C., Hansen T., Herder C., Hitman G. A., Hughes T. E., Isomaa B., Jackson A. U., Jørgensen T., Kong A., Kubalanza K., Kuruvilla F. G., Kuusisto J., Langenberg C., Lango H., Lauritzen T., Li Y., Lindgren C. M., Lyssenko V., Marvelle A. F., Meisinger C., Midthjell K., Mohlke K. L., Morken M. A., Morris A. D., Narisu N., Nilsson P., Owen K. R., Palmer C. N., Payne F., Perry J. R., Pettersen E., Platou C., Prokopenko I., Qi L., Qin L., Rayner N. W., Rees M., Roix J. J., Sandbaek A., Shields B., Sjögren M., Steinthorsdottir V., Stringham H. M., Swift A. J., Thorleifsson G., Thorsteinsdottir U., Timpson N. J., Tuomi T., Tuomilehto J., Walker M., Watanabe R. M., Weedon M. N., Willer C. J., Illig T., Hveem K., Hu F. B., Laakso M., Stefansson K., Pedersen O., Wareham N. J., Barroso I., Hattersley A. T., Collins F. S., Groop L., McCarthy M. I., Boehnke M., Altshuler D. Meta-analysis of genome-wide association data and large-scale replication identifies additional susceptibility loci for type 2 diabetes. Genet. 2008;40:638–645.
CrossRef - Saxena R., Elbers C. C., Guo Y., Peter I., Gaunt T. R., Mega J. L., Lanktree M. B., Tare A., Castillo B. A., Li Y. R., Johnson T.,Bruinenberg M., Gilbert D. D., Rajagopalan R., Voight B. F., Balasubramanyam A., Barnard J., Bauer F., Baumert J., Bhangale T., Böhm B. O., Braund P. S., Burton P. R., Chandrupatla H. R., Clarke R., Cooper-DeHoff M., Crook E. D., Davey-S. G., Day I. N., de B. A., de Groot M. C., Drenos F., Ferguson J., Fox C. S., Furlong C. E., Gibson Q., Gieger C., Gilhuijs-Pederson L. A.,Glessner J. T., Goel A., Gong Y., Grant S. F., Grobbee D. E., Hastie C., Humphries S. E., Kim C. E., Kivimaki M., Kleber M., Meisinger C., Kumari M., Langaee T. Y., Lawlor D. A., Li M., Lobmeyer M. T., Maitland-van der Zee A. H., Meijs M. F., Molony C. M., Morrow D. A., Murugesan G., Musani S. K., Nelson C. P., Newhouse S. J., O’Connell J. R., Padmanabhan S., Palmen J., Patel S. R., Pepine C. J., Pettinger M., Price T. S., Rafelt S., Ranchalis J., Rasheed A., Rosenthal E., Ruczinski I., Shah S., Shen H., Silbernagel G., Smith E. N., Spijkerman A. W., Stanton A., Steffes M. W., Thorand B., Trip M., van der H. P., van der A. D. L., van Iperen E. P., van S. J., van Vliet-Ostaptchouk J. V., Verweij N., Wolffenbuttel B. H., Young T., Zafarmand M. H., Zmuda J. M., Boehnke M., Altshuler D., McCarthy M., Kao W. H., Pankow J. S., Cappola T. P., Sever P., Poulter N., Caulfield M., Dominiczak A., Shields D. C., Bhatt D. L., Zhang L., Curtis S. P., Danesh J., Casas J. P., van der Schouw Y. T., Onland-Moret N. C., Doevendans P. A., Dorn G. W., Farrall M., FitzGerald G. A., Hamsten A., Hegele R., Hingorani A. D., Hofker M. H., Huggins G. S., Illig T., Jarvik G. P., Johnson J. A., Klungel O. H., Knowler W. C., Koenig W., März W., Meigs J. B., Melander O., Munroe P. B., Mitchell B. D., Bielinski S. J., Rader D. J., Reilly M. P., Rich S. S., Rotter J. I., Saleheen D., Samani N. J., Schadt E. E., Shuldiner A. R., Silverstein R., Kottke M. K., Talmud P. J., Watkins H., Asselbergs F. W., de Bakker P. I., McCaffery J., Wijmenga C., Sabatine M. S., Wilson J. G., Reiner A., Bowden D. W., Hakonarson H., Siscovick D. S., Keating B. J. Large-scale gene-centric meta-analysis across 39 studies identifies type 2 diabetes loci. Am. J. Hum. Genet. 2012; 90:410-425.
CrossRef - Dupuis J., Langenberg C., Prokopenko I., Saxena R., Soranzo N., Jackson A. U., Wheeler E., Glazer N. L., Bouatia-Naji N., Gloyn A. L., Lindgren C. M., Mägi R., Morris A. P., Randall J., Johnson T., Elliott P., Rybin D., Thorleifsson G., Steinthorsdottir V., Henneman P., Grallert H., Dehghan A., Hottenga J. J., Franklin C. S., Navarro P., Song K., Goel A., Perry J. R., Egan J. M., Lajunen T., Grarup N., Sparsø T., Doney A., Voight B. F., Stringham H. M., Li M., Kanoni S., Shrader P., Cavalcanti-Proença C., Kumari M., Qi L., Timpson N. J., Gieger C., Zabena C., Rocheleau G., Ingelsson E., An P., O’Connell J., Luan J., Elliott A., McCarroll S. A., Payne F., Roccasecca R. M., Pattou F., Sethupathy P., Ardlie K., Ariyurek Y., Balkau B., Barter P., Beilby J. P., Ben-Shlomo Y., Benediktsson R., Bennett A. J., Bergmann S., Bochud M., Boerwinkle E., Bonnefond A., Bonnycastle L.L., Borch-Johnsen K., Böttcher Y., Brunner E., Bumpstead S. J., Charpentier G., Chen Y. D., Chines P., Clarke R., Coin L. J., Cooper M. N., Cornelis M., Crawford G., Crisponi L., Day I. N., de Geus E. J., Delplanque J., Dina C., Erdos M. R., Fedson A. C., Fischer-Rosinsky A, Forouhi N. G., Fox C. S., Frants R., Franzosi M. G., Galan P., Goodarzi M. O., Graessler J., Groves C. J., Grundy S., Gwilliam R., Gyllensten U., Hadjadj S., Hallmans G., Hammond N., Han X., Hartikainen A. L., Hassanali N., Hayward C., Heath S. C., Hercberg S., Herder C., Hicks A. A., Hillman D. R., Hingorani A. D., Hofman A., Hui J., Hung J., Isomaa B., Johnson P. R., Jørgensen T., Jula A., Kaakinen M., Kaprio J., Kesaniemi Y. A., Kivimaki M., Knight B., Koskinen S., Kovacs P., Kyvik K. O., Lathrop G. M., Lawlor D. A., Le Bacquer O., Lecoeur C., Li Y., Lyssenko V., Mahley R., Mangino M., Manning A. K., Martínez-Larrad M. T., McAteer J. B., McCulloch L. J., McPherson R., Meisinger C., Melzer D., Meyre D., Mitchell B. D., Morken M. A., Mukherjee S., Naitza S., Narisu N., Neville M. J., Oostra B. A., Orrù M., Pakyz R., Palmer C. N., Paolisso G., Pattaro C., Pearson D., Peden J. F., Pedersen N. L., Perola M., Pfeiffer A. F., Pichler I., Polasek O., Posthuma D., Potter S. C., Pouta A., Province M. A., Psaty B. M., Rathmann W., Rayner N. W., Rice K., Ripatti S., Rivadeneira F., Roden M., Rolandsson O., Sandbaek A., Sandhu M., Sanna S., Sayer A. A., Scheet P., Scott L. J., Seedorf U., Sharp S. J., Shields B., Sigurethsson G., Sijbrands E. J., Silveira A., Simpson L., Singleton A., Smith N. L., Sovio U., Swift A., Syddall H., Syvänen A. C., Tanaka T., Thorand B., Tichet J., Tönjes A., Tuomi T., Uitterlinden A. G., van Dijk K. W., van H. M, Varma D., Visvikis S. S., Vitart V., Vogelzangs N., Waeber G., Wagner P. J., Walley A., Walters G. B., Ward K. L., Watkins H., Weedon M. N., Wild S. H., Willemsen G., Witteman J. C., Yarnell J. W., Zeggini E., Zelenika D., Zethelius B., Zhai G., Zhao J. H., Zillikens M. C., Borecki I. B., Loos R. J., Meneton P., Magnusson P. K., Nathan D. M., Williams G. H., Hattersley A. T., Silander K., Salomaa V., Smith G. D., Bornstein S. R., Schwarz P., Spranger J., Karpe F., Shuldiner A. R., Cooper C., Dedoussis G. V., Serrano-Ríos M., Morris A. D., Lind L., Palmer L. J., Hu F. B., Franks P. W., Ebrahim S., Marmot M., Kao W. H., Pankow J. S., Sampson M. J., Kuusisto J., Laakso M., Hansen T., Pedersen O., Pramstaller P. P., Wichmann H. E., Illig T., Rudan I., Wright A. F., Stumvoll M., Campbell H., Wilson J. F., Bergman R. N., Buchanan T. A., Collins F. S., Mohlke K. L., Tuomilehto J., Valle T. T., Altshuler D., Rotter J. I., Siscovick D. S., Penninx B. W., Boomsma D. I., Deloukas P., Spector T. D., Frayling T. M., Ferrucci L., Kong A,. Thorsteinsdottir U., Stefansson K., van Duijn C. M., Aulchenko Y. S., Cao A., Scuteri A., Schlessinger D., Uda M., Ruokonen A., Jarvelin M. R., Waterworth D. M., Vollenweider P., Peltonen L., Mooser V., Abecasis G. R., Wareham N. J., Sladek R., Froguel P., Watanabe R. M., Meigs J. B., Groop L., Boehnke M., McCarthy M. I., Florez J. C., Barroso I. New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Genet. 2010;42:105–116.
- Morris A. P., Voight B. F., Teslovich T. M., Ferreira T., Segrè A.V., Steinthorsdottir V., Strawbridge R. J., Khan H., Grallert H., Mahajan A., Prokopenko I., Kang H. M., Dina C., Esko T., Fraser R. M., Kanoni S., Kumar A., Lagou V., Langenberg C., Luan J., Lindgren C. M., Müller N. M., Pechlivanis S., Rayner N. W., Scott L. J., Wiltshire S., Yengo L., Kinnunen L., Rossin E. J., Raychaudhuri S., Johnson A. D., Dimas A. S., Loos R. J., Vedantam S., Chen H., Florez C., Fox C., Liu C. T., Rybin D., Couper D. J.,Kao W. H., Li M., Cornelis M. C., Kraft P., Sun Q., van Dam R. M., Stringham H. M., Chines P. S., Fischer K., Fontanillas P, Holmen O. L.,Hunt S. E., Jackson A. U., Kong A., Lawrence R., Meyer J., Perry J. R., Platou C. G., Potter S., Rehnberg E., Robertson N., Sivapalaratnam S., Stančáková A., Stirrups K., Thorleifsson G., Tikkanen E., Wood A. R., Almgren P., Atalay M., Benediktsson R., Bonnycastle L. L., Burtt N., Carey J., Charpentier G., Crenshaw A. T., Doney A. S., Dorkhan M., Edkins S., Emilsson V., Eury E., Forsen T., Gertow K., Gigante B., Grant G. B., Groves C. J., Guiducci C., Herder C., Hreidarsson A. B., Hui J., James A., Jonsson A., Rathmann W., Klopp N., Kravic J., Krjutškov K., Langford C., Leander K., Lindholm E., Lobbens S, Männistö S., Mirza G., Mühleisen T. W., Musk B., Parkin M., Rallidis L., Saramies J., Sennblad B., Shah S., Sigurðsson G., Silveira A., Steinbach G., Thorand B., Trakalo J., Veglia F., Wennauer R., Winckler W., Zabaneh D., Campbell H., van Duijn C., Uitterlinden A. G., Hofman A., Sijbrands E., Abecasis G. R., Owen K. R., Zeggini E., Trip M. D., Forouhi N. G., Syvänen A. C., Eriksson J. G., Peltonen L., Nöthen M. M., Balkau B., Palmer C. N., Lyssenko V., Tuomi T., Isomaa B., Hunter D. J., Qi L., Shuldiner A. R., Roden M., Barroso I., Wilsgaard T., Beilby J., Hovingh K., Price J. F., Wilson J. F., Rauramaa R., Lakka T. A., Lind L., Dedoussis G., Njølstad I., Pedersen N. L., Khaw K. T., Wareham N. J., Keinanen-Kiukaanniemi S. M., Saaristo T. E., Korpi H. E., Saltevo J., Laakso M., Kuusisto J., Metspalu A., Collins F. S., Mohlke K. L., Bergman R. N., Tuomilehto J., Boehm B. O., Gieger C., Hveem K., Cauchi S., Froguel P., Baldassarre D., Tremoli E., Humphries S. E., Saleheen D., Danesh J., Ingelsson E., Ripatti S., Salomaa V., Erbel R., Jöckel K. H., Moebus S., Peters A., Illig T., de Faire U., Hamsten A., Morris A. D., Donnelly P. J., Frayling T. M., Hattersley A. T., Boerwinkle E., Melander O.,Kathiresan S., Nilsson P. M., Deloukas P., Thorsteinsdottir U., Groop L. C., Stefansson K., Hu F., Pankow J. S., Dupuis J., Meigs J. B., Altshuler D., Boehnke M., McCarthy M. I. Large-scale association analysis provides insights into the genetic architecture and pathophysiology of type 2 diabetes. Nat. Genet. 2012;44:981-990.
CrossRef - Harder M. N., Ribel-M. R., Justesen J. M., Sparsø T., Andersson E. A., Grarup N., Jørgensen T., Linneberg A., Hansen T., Pedersen O. Type 2 diabetes risk alleles near BCAR1 and in ANK1 associate with decreased β-cell function whereas risk alleles near ANKRD55 and GRB14 associate with decreased insulin sensitivity in the Danish Inter99 cohort. Clin. Endocrinol. Metab. 2013;98:E801-E806.
CrossRef - Imamura M., Maeda S., Yamauchi T., Hara K., Yasuda K., Morizono T., Takahashi A., Horikoshi M., Nakamura M., Fujita H., Tsunoda T., Kubo M., Watada H., Maegawa H., Okada- Iwabu M., Iwabu M., Shojima N., Ohshige T., Omori S., Iwata M., Hirose H., Kaku K., Ito C., Tanaka Y., Tobe K., Kashiwagi A., Kawamori R., Kasuga M., Kamatani N., Nakamura Y., Kadowaki T. A single-nucleotide polymorphism in ANK1 is associated with susceptibility to type 2 diabetes in Japanese populations. Mol. Genet. 2012;21:3042-3049.
- Perry J. R., Voight B. F., Yengo L., Amin N., Dupuis J., Ganser M., Grallert H., Navarro P., Li M., Qi L., Steinthorsdottir V., Scott R. A., Almgren P., Arking D. E., Aulchenko Y., Balkau B., Benediktsson R., Bergman R. N., Boerwinkle E., Bonnycastle L., Burtt N. P., Campbell H., Charpentier G., Collins F. S., Gieger C., Green T., Hadjadj S., Hattersley A. T., Herder C., Hofman A., Johnson A. D., Kottgen A., Kraft P., Labrune Y., Langenberg C., Manning A. K., Mohlke K. L., Morris A. P., Oostra B., Pankow J., Petersen A. K., Pramstaller P. P., Prokopenko I., Rathmann W., Rayner W., Roden M., Rudan I., Rybin D., Scott L. J., Sigurdsson G., Sladek R., Thorleifsson G., Thorsteinsdottir U., Tuomilehto J., Uitterlinden A. G., Vivequin S., Weedon M. N., Wright A. F., Hu F. B., Illig T., Kao L., Meigs J. B., Wilson J. F., Stefansson K., van Duijn C., Altschuler D., Morris A. D., Boehnke M., McCarthy M. I., Froguel P., Palmer C. N., Wareham N. J., Groop L., Frayling T. M., Cauchi S. Stratifying type 2 diabetes cases by BMI identifies genetic risk variants in LAMA1 and enrichment for risk variants in lean compared to obese cases. Genet. 2012;8:e1002741.
CrossRef - Weedon M. N., Schwarz P. E., Horikawa Y., Iwasaki N., Illig T.,Holle R., Rathmann W., Selisko T., Schulze J., Owen K. R., Evans J., Del Bosque-Plata L., Hitman G., Walker M., Levy J. C., Sampson M., Bell G. I., McCarthy M. I., Hattersley A. T., Frayling T.M.. Meta-analysis and a large association study confirm a role for calpain-10 variation in type 2 diabetes susceptibility. J. Hum. Genet. 2003;73:1208-1212.
CrossRef - Jensen D. P., Urhammer S. A., Eiberg H., Borch-Johnsen K., Jørgensen T., Hansen T., Pedersen O. Variation in CAPN10 in relation to type 2 diabetes, obesity and quantitative metabolic traits: studies in 6018 whites. Genet. Metab. 2006;89:360-367.
CrossRef - Tsuchiya T., Schwarz P. E., Bosque-Plata L. D., Hayes M. G., Dina C., Froguel P., Towers G. W., Fischer S., Temelkova Kurktschiev T., Rietzsch H., Graessler J., Vcelák J., Palyzová D., Selisko T., Bendlová B., Schulze J., Julius U., Hanefeld M., Weedon M. N., Evans J. C., Frayling T. M., Hattersley A. T., Orho- Melander M., Groop L., Malecki M. T., Hansen T., Pedersen O., Fingerlin T. E., Boehnke M., Hanis C. L., Cox N. J., Bell G. I. Association of the calpain-10 gene with type 2 diabetes in Europeans:results of pooled and meta-analyses. Genet. Metab. 2006;89:174-184.
CrossRef - Song Y., Niu T., Manson J. E., Kwiatkowski D. J., Liu S. Are variants in the CAPN10 gene related to risk of type 2 diabetes? A quantitative assessment of population and family-based association studies. J. Hum. Genet. 2004;74:208-222.
CrossRef - Saxena R., Voight B. F., Lyssenko V., Burtt N. P., de Bakker P. I.,Chen H., Roix J. J., Kathiresan S., Hirschhorn J. N., Daly M. J.,Hughes T. E., Groop L., Altshuler D., Almgren P., Florez C.,Meyer J., Ardlie K., Boström K. B., Isomaa B., Lettre G.,Lindblad U., Lyon H. N., Melander O., Newton-Cheh C., Nilsson P., Orho-Melander M., Råstam L., Speliotes E. K., Taskinen M. R., Tuomi T., Guiducci C., Berglund A., Carlson J., Gianniny L.,Hackett R., Hall L., Holmkvist J., Laurila E., Sjögren M., Sterner M., Surti A., Svensson M., Svensson M., Tewhey R., Blumenstiel B., Parkin M., Defelice M., Barry R., Brodeur W., Camarata J.,Chia N., Fava M., Gibbons J., Handsaker B., Healy C., Nguyen K., Gates C., Sougnez C., Gage D., Nizzari M., Gabriel S. B., Chirn G. W., Ma Q., Parikh H., Richardson D., Ricke D., Purcell S. Genome-wide association analysis identifies loci for type 2 diabetes and triglyceride levels. Science. 2007;316:1331-1336.
CrossRef - Scott L. J., Mohlke K. L., Bonnycastle L. L., Willer C. J., Li Y., Duren W. L., Erdos M. R., Stringham H. M., Chines P. S., Jackson A. U.,Prokunina-Olsson L., Ding C. J., Swift A. J., Narisu N., Hu T.,Pruim R., Xiao R., Li X. Y., Conneely K. N., Riebow N. L., Sprau A. G., Tong M., White P. P., Hetrick K. N., Barnhart M. W., Bark C. W., Goldstein J. L., Watkins L., Xiang F., Saramies J., Buchanan T. A., Watanabe R. M., Valle T. T., Kinnunen L., Abecasis G. R.,Pugh E. W., Doheny K. F., Bergman R. N., Tuomilehto J., Collins F. S., Boehnke M. A genome-wide association study of type 2 diabetes in Finns detects multiple susceptibility variants. Science. 2007;316:1341-1345.
CrossRef - Steinthorsdottir V., Thorleifsson G., Reynisdottir I., Benediktsson R., Jonsdottir T., Walters G. B., Styrkarsdottir U.,Gretarsdottir S., Emilsson V., Ghosh S., Baker A., Snorradottir S., Bjarnason H., Ng M. C., Hansen T., Bagger Y., Wilensky R. L., Reilly M. P., Adeyemo A., Chen Y., Zhou J., Gudnason V., Chen G., Huang H., Lashley K., Doumatey A., So W. Y., Ma R. C., Andersen G., Borch-Johnsen K., Jorgensen T., van Vliet-Ostaptchouk J. V., Hofker M. H., Wijmenga C., Christiansen C., Rader D. J., Rotimi C., Gurney M., Chan J. C., Pedersen O., Sigurdsson G., Gulcher J. R., Thorsteinsdottir U., Kong A., Stefansson K. Avariant in CDKAL1 influences insulin response and risk of type 2 diabetes. Genet. 2007;39:770-775.
- Zeggini E., Weedon M. N., Lindgren C. M., Frayling T. M., Elliott K. S., Lango H., Timpson N. J., Perry J. R., Rayner N. W., Freathy R. M., Barrett J. C., Shields B., Morris A. P., Ellard S., Groves C. J.,Harries L. W., Marchini J. L., Owen K. R., Knight B., Cardon L. R.,Walker M., Hitman G. A., Morris A. D., Doney A. S., McCarthy M. I., Hattersley A. T. Replication of genome-wide association signals in UK samples reveals risk loci for type 2 diabetes.Science. 2007;316:1336-1341.
CrossRef - Rong Y., Bao W., Rong S., Fang M., Wang D., Yao P., Hu F. B., Liu L. Hemochromatosis gene (HFE) polymorphisms and risk of type 2 diabetes mellitus: a meta-analysis. J. Epidemiol. 2012;176:461-472 .
CrossRef - Sladek R., Rocheleau G., Rung J., Dina C., Shen L., Serre D., Boutin P., Vincent D., Belisle A., Hadjadj S., Balkau B., Heude B., Charpentier G., Hudson T. J., Montpetit A., Pshezhetsky A. V., Prentki M.,Posner B. I., Balding D. J., Meyre D., Polychronakos C., Froguel P. A genome-wide association study identifies novel risk loci fortype 2 diabetes. Nature 2007;445:881-885.
CrossRef - Almind K., Bjørbaek C., Vestergaard H., Hansen T., Echwald S.,Pedersen O. Aminoacid polymorphisms of insulin receptor substrate-1 in non-insulin-dependent diabetes mellitus. 1993;342:828-832.
- Deeb S. S., Fajas L., Nemoto M., Pihlajamäki J., Mykkänen L., Kuusisto J., Laakso M., Fujimoto W., Auwerx J. A Pro12Ala substitution in PPARgamma2 associated with decreased receptor activity, lower body mass index and improved insulin sensitivity. Genet. 1998;20:284-287.
- Kong A., Steinthorsdottir V., Masson G., Thorleifsson G.,Sulem P., Besenbacher S., Jonasdottir A., Sigurdsson A., Kristinsson K. T., Jonasdottir A., Frigge M. L., Gylfason A., Olason P. I., Gudjonsson S. A., Sverrisson S., Stacey S. N., Sigurgeirsson B., Benediktsdottir K. R., Sigurdsson H., Jonsson T., Benediktsson R., Olafsson J. H., Johannsson O. T., Hreidarsson A. B., Sigurdsson G., Ferguson-Smith A. C., Gudbjartsson D. F., Thorsteinsdottir U., Stefansson K. Parental origin of sequence variants associated with complex diseases. Nature. 2009;462:868-874.
CrossRef - Dixit P. P., Londhe J. S., Ghaskadbi S. S and Devasagayam T. P. A. Antidiabetic and related beneficial properties of Indian medicinal plants, in Herbal Drug Research- A twenty first century perspective, eds. By Sharma R. K and Arora R., Jaypee brothers medical publishers (New Delhi, India) Limited. 2006;377–386.
- Ahmad S. Lokaj chikitsay vesaja udvid (Traditional treatments and medicinal plants). Dhaka: Anupam Prokashni. 2012;1-775.
- Alam M. J. Traditional medicine in Bangladesh. Dhaka:Asiatic Society of Bangladesh. 2007;1-311.
- Ghani A. Medicinal plants of Bangladesh. Dhaka: Asiatic Society of Bangladesh. 1998.
- Yusuf M., Begum J., Hoque M. N., Choudhury J. U. Medicinal plants of Bangladesh—revised and enlarged. Chittagong: Bangladesh Coun. Sci. Ind. Res. Lab. 2009.
- Trivedi N. P., Rawal U. M. Hepatoprotective and antioxidant property of Andrographis paniculata (Nees) in BHC induced liver damage in mice. Indian J. Exp. Biol. 2001;39(1):41-46.
- Kumar R.,Pate K. D.,Prasad K. S.,Sairam K.,Hemalatha S. Antidiabetic activity of alcoholic leaves extract of Alangium lamarckii Thwaites on streptozotocin nicotinamide induced type 2 diabetic rats. Asian Pac. J. Tropical Med. 2011;904-909.
CrossRef - Kumar D.,Kumar S., Kohli S., Arya R.,Gupta J. Antidiabetic activity of methanolic bark extract of Albizia odoratissima Benth in alloxan induced diabetic albino mice. Asian Pac. J. Trop. Med. 2011;900-903.
CrossRef - Sancheti S., Sancheti S., Seo S. Y. Antidiabetic and antiacetylcholinesterase effects of ethyl acetate fraction of Chaenomeles sinensis (Thouin) Koehne fruits in streptozotocin induced diabetic rats. Toxicol. Pathol. 2011;65(1-2):55-60.
CrossRef - Wadood A.,Wadood N and Shah S. A. Effects of Acacia arabica and Caralluma edulis on blood glucose levels on normal and alloxan diabetic rabbits. J.Pakistan. Med. Assoc. 1989;39:208–212.
- Karunanayake H., Welihinda J., Sirimanne S. R and Sinnadorai G. the Oral hypoglycemic activity of some medicinal plants of Sri Lanka. J.Ethnopharmacol. 11:223–231. 19.84.
- Ramos R. R., Saenz J. L. F and Alarcon-Aguilar F. J. Antihyperglycemic effect of some edible plants. J. Ethnopharmacol. 1995;48:25–32.
CrossRef - Kumari K., Mathew B. C and Augusti K. T. Antidiabetic and hypolipidaemic effects of S-methyl cysteine sulfoxide, isolated from Allium cepa Linn. Ind. J. Biochem. Biophys. 1995;32:49–54.
- Mathew P. T and Augusti K. T. Hypoglycemic effects of onion, Allium cepa Linn. on diabetes mellitus- a preliminary report. Ind. J. Physiol. Pharmacol. 1975;19:213–217.
- Al-Awadi F. M and Gumaa K. A. Studies on the activity of individual plants of an antidiabetic plant mixture. Acta Diabetologica 1987;24:37–41.
CrossRef - Davis R. H and Maro N. P. Aloe vera and gibberellins, Anti-inflammatory activity in diabetes. J. Am. Pediat. Med. Assoc. 1989;79:24–26.
CrossRef - Jeppesen P. B., Gregersen S., Alstrup K. K., Hermansen K. Stevioside induces antihyperglycaemic, insulinotropic and glucagonostatic effects in vivo: studies in the diabetic Goto-Kakizaki (GK) rats. Phytomedicine. 2002;9(1):9-14.
CrossRef - Jeppesen P. B., Gregersen S., Poulsen C. R., Hermansen K. Stevioside acts directly on pancreatic beta cells to secrete insulin: actions independent of cyclic adenosine monophosphate and adenosine triphosphate-sensitive K+-channel activity. Metabolism. 2000;49(2):208-214.
CrossRef - Devendra D., Liu E., Eisenbarth G. S. Type 1 diabetes: recent developments. BMJ. 2004;328:750-754.
CrossRef - Dabelea D., Mayer-Davis E. J., Saydah S., Imperatore G., Linder B., Divers J., Bell R., Badaru A., Talton J. W., Crume T., Liese A. D.,Merchant A. T., Lawrence J. M., Reynolds K., Dolan L., Liu L. L., Hamman R. F. Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA. 2014;311:1778-1786.
CrossRef - Vermeulen I., Weets I., Asanghanwa M., Ruige J., Van Gaal L.,Mathieu C., Keymeulen B., Lampasona V., Wenzlau J. M., Hutton J. C.,Pipeleers D. G., Gorus F. K. Contribution of antibodies against IA-2β and zinc transporter 8 to classification of diabetes diagnosed under 40 years of age. Diabetes Care. 2011;34:1760-1765.
CrossRef - Couper J., Donaghue K. C. Phases of diabetes in children and adolescents. Pediatr Diabetes. 2009;10(12):13-16.
CrossRef - Ginsberg-Fellner F., Witt M. E., Fedun B., Taub F., Dobersen M. J.,McEvoy R. C., Cooper L. Z., Notkins A. L., Rubinstein P. Diabetes mellitus and autoimmunity in patients with the congenital rubella syndrome. Infect. Dis. 1985;7(1):S170-S176.
CrossRef - McIntosh E. D., Menser M. A. A fifty-year follow-up of congenital rubella. Lancet. 1992;340:414-415.
CrossRef - Stene L. C., Oikarinen S., Hyöty H., Barriga K. J., Norris J. M., Klingensmith G., Hutton J. C., Erlich H. A., Eisenbarth G. S., Rewers M. Enterovirus infection and progression from islet autoimmunity to type 1 diabetes: the Diabetes and Autoimmunity Study in the Young (DAISY). Diabetes. 2010;59:3174-3180.
CrossRef - Imagawa A., Hanafusa T. Fulminant type 1 diabetes–an important subtype in East Asia. Diabetes Metab. Res. Rev. 2011;27: 959-964.
CrossRef - Shibasaki S., Imagawa A., Hanafusa T. Fulminant type 1 diabetes mellitus: a new class of type 1 diabetes. Exp. Med. Biol. 2012;771: 20-23.
- Imagawa A., Hanafusa T. Fulminant type 1 diabetes mellitus. Endocr J. 2006;53:577-584.
CrossRef - Shimizu I., Makino H., Imagawa A.,Iwahashi H., Uchigata Y.,Kanatsuka A., Kawasaki E., Kobayashi T., Shimada A,Maruyama T,Hanafusa T. Clinical and immunogenetic characteristics of fulminant type 1 diabetes associated with pregnancy. J Clin Endocrinol Metab. 2006;91:471-476.
CrossRef - International Diabetes Federation. IDF Diabetes Atlas. 8th ed.Brussels, Belgium: International Diabetes Federation, 2017.
- Galloway JA: Diabetes Mellitus. 9th ed. Indianapolis, Ind., Eli Lilly and Company. 1988.
- Daneman D., Drash A. L., Lobes L. A., Becker D. J., Baker L. M., Travis L. B. Progressive retinopathy with improved control in diabetic dwarfism (Mauriac’s syndrome). Diabetes Care. 1981;4:360–365.
CrossRef - Defeat Diabetes, Foundation: History of diabetes in timeline. Available from http://www.defeatdiabetes.org/about_diabetes/ text.asp?id=Diabetes_Timeline. Accessed 4 December. 2013.
- Medscape. FDA rejects Novo Nordisk’s insulin degludec [article online]. 11 February 2013. Available from http://www. medscape.com/viewarticle/779077. Accessed 2 January. 2014.
- White J. R. Overview of the medications used to treat type 2 diabetes. In Medications for the Treatment of Diabetes. White J. R., Campbell R. K., Eds. Alexandria. Va., American Diabetes Association. 2008;5–15
- Frank E., Nothnamm M., Wagner A. Über synthetische dargestellte Korper mit Insulinartiger Wirkung auf den normallen und diabetisched Organismus. Klin Wchnschr. 1926;5:2011.
CrossRef - Alberti K. G. M. M., Zimmet P., Defronzo R. A. International Textbook of Diabetes Mellitus. 2nd ed. New York, John Wiley & Sons. 1997.
- Levine R. Sulfonylureas: background and development of the field. Diabetes Care. 1984;7(1):3–7.
- Quianzon CCL, Cheikh I: History of current non-insulin medications for diabetes mellitus. J Community Hosp Intern Med Perspect. Published online 15 October 2012.
CrossRef - Aiello L. P., Gardner T. W., King G. L et al Diabetic retinopathy. Diabetes Care. 1998;21:143–56.
CrossRef - Chen Y. D., Reaven G. M. Insulin resistance and atherosclerosis. Diabetes Rev. 1997;5:331–43.
- Ritz E., Orth S. R. Nephropathy in patients with type 2 diabetes. N Engl J Med. 1999;341:1127–33.
CrossRef - Grundy S. M., Benjamin I. J., Burke G. L et al Diabetes and cardiovascular disease: A statement for healthcare professionals from the American Heart Association. Circulation. 1999;100:1134–46.
CrossRef - Consensus development conference on diabetic foot wound care, (American Diabetes Association, 7–8 April 1999, Boston). Diabetes Care. 1999;22:1354–60.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





