How to Cite | Publication History | PlumX Article Matrix
Shadi Barzanjeh1, Hosein Mostafavi1, Mohsen Alipour1,Mehdi Eskandari1*and Saeed Ghanbarzadeh2
1Department of Medicinal Physiology and Pharmacology, Faculty of Medicine, Zanjan University of Medical Sciences, Zanjan, Iran.
2Department of Pharmaceutics, Faculty of Pharmacy, Zanjan University of Medical Sciences, Zanjan, Iran.
Corresponding Author E-mail: Mehdiesk@zums.ac.ir.
DOI : http://dx.doi.org/10.13005/bbra/2336
ABSTRACT: The purposes of this investigation were toexamine the effects of exercise and smoking throughout pregnancy on placentagrowth and vascularization, human chorionic gonadotropic hormone (HCG) levelandbirthweight. Rats in group 1 were selected as control group and were not in exposure of smoke and not exercised. Rats in group 2 were exercised for 5 days in week for 3 weeks. Rats in group 3 were in the cigarette smoke exposurewithout exercise. Finally, rats in group 4 were forced to exercise and were in the exposure of cigarette smoke, simultaneously.Six rats from each group were killed after 3 weeks of pregnancy and the number of blood vessels, placenta decidual thickness, and HCG level were analyzed. Birthweight and weight of offspringafter 60 days in each group were also evaluated. Results showed that the number of blood vessels and HCG level were decrease in animals which exposed to smoke and exercise (P<0.05),however, the spatula decidua thickness did not changed significantly. The offspring birthweight in group 4 was decreased significantly (P<0.001) in comparison with control group. Accordingly, the offspring birthweight in group 3 and 4 was decrease significantly compared to group 2 (P<0.01). On the otherhand, the weight of offspring in day 60 in group 3 was less than group 4. In conclusion, both exercise and smoking have negative effect on blood vessels, HCG level, and offspringbirthweight, however, there is synergic effect when rats were in exposure of smoke and exercise, simultaneously.
KEYWORDS: CigaretteSmoke; Exercise; Pregnancy; Placenta; Birthweight
Download this article as:| Copy the following to cite this article: Barzanjeh S, Mostafavi H, Alipour M, Eskandari M, Ghanbarzadeh S. Effect of Cigarette Smoke Exposure and Exercise on Abortion, Placental Vascularization, Human Chronic Gonadotropin Hormone and Birthweight. Biotech Res Asia 2016;13(3). |
| Copy the following to cite this URL: Barzanjeh S, Mostafavi H, Alipour M, Eskandari M, Ghanbarzadeh S. Effect of Cigarette Smoke Exposure and Exercise on Abortion, Placental Vascularization, Human Chronic Gonadotropin Hormone and Birthweight. Biotech Res Asia 2016;13(3). Available from: https://www.biotech-asia.org/?p=16129 |
Introduction
Cigarette smoking in the around the worldis a critical challenge for women and children’shealth. Cigarette smoke contains about 4800compounds of which at least 60 are classified as carcinogens.Cigarette smoking causes 30% of all cancer deaths in developed countries. It is estimated that cigarette smoking kills over 1000000 people each year by causing lung cancer and many other neoplasmas. In addition to lung cancer, cigarette smoking is an important cause of esophageal, oral, oropharyngeal, hypopharyngeal, and laryngeal cancers as well as pancreatic cancer, bladder cancer, liver, colon, nose, and myeloid leukemia, and cancer of the renal [1-4].Some other components have adverse effects on the cardiovascular, respiratory, reproductive, and nervous systems[5-9]. TheWorld Health Organization reports that the smokingrates for women (≥15-year old) range between 7-24%in the U.S., Europe, and Australia.It has been widely reportedthat smoking during pregnancy or secondhand smoke, as involuntarysmoking,have negative reproductive effects including reducedfetal growth,low birth weight,premature delivery, spontaneous abortion, placental abruption,perinatal mortality and ectopic pregnancyas well as increased risk of fetal mortality.Furthermore, maternal smoking accounts for an estimated 20–30% of alllow birthweight babies, as well as for 14% of allpreterm deliveries, and some 10% of all infant death [10-13]. In several animal models and thehuman, regular maternal exercise reduces birthweight.Recent findings suggested that regular, sustained, moderate to high intensity exerciseduring pregnancy might changevessel growth, blood flow, surface area and the configuration of the villous tree. Furthermore, theconcern was heightened by the other reportsindicating to reduction in all villous dimensions inplacentae from human pregnancies at high altitude, and morphometric outcome is similar to that seen with regular exercise throughoutpregnancy[14-17].Thus, the current study was undertaken to determine if exercise can attenuate the negative effect of cigarette smoke exposure during pregnancy.
Methods
Animals
All animal experiments were conducted according to the Guide for Care and Use of Laboratory Animals of Zanjan University of Medical Sciences, Zanjan-Iran (National Institutes of Health Publication No 85-23, revised 1985). Thirty two adult female Wistar rats (weighing 200-250 g, aged 8 weeks) were divided into four groups (n=8). In group 1 (Controlgroup) animals were not exposed tosmoke and not exercised. To investigate the effect of smoke and exercise, animals were exposed tosmoke or forced to exercise5 days in a week for 3 weeks, respectively. Rats in group 4 were both in exposed to smoke and exercised. Animals had free access to food and water under standard lighting conditions (12-h light: 12-h darkness). After 3 weeks 6 rats from each group were sacrificed and their spatula decidual thickness, HCG level (using enzyme-linked immunosorbent assay, ELISA)and the number of blood vessels and abortion were evaluated. The offspring birthweight and their weight after 60 days of remained rats were also measured.
Tissues from each group were fixed in formaldehyde, embedded in paraffinwax, sectioned at 6 micrometer and stained usinghematoxyline and eosin (H&E) technique.After preparation of the sections, 3 slides werechosen randomly from each sample and at least fourfields of view were measured from each slide. Thefollowing blood vessels number and the spatula decidual thickness were measured ineach field of view.
Statistical Analysis
Data are expressed as mean ± SD and analyzed using the student’s t-test and ANOVA where necessary.P˂ 0.05was consideredstatistically significant.
Results and discussion
Effect of smoke and exercise on HCG level
Fig. 1 represents the effect of exposure to smoke and exercise on the HCG level in rats in four investigated groups. As shown in both smoke exposed and smoke exposed-exercised groups the level of HCG were decreased, however, the decrease in HCG level was significant in smoke exposed-exercised group (P<0.05). Furthermore, results indicated that the level of HCG did not decrease in exercised rat which were not in exposure of smoke.
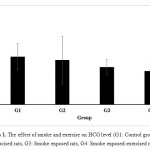 |
Figure 1: The effect of smoke and exercise on HCG level (G1: Control group, G2: Exercised rats, G3: Smoke exposed rats, G4: Smoke exposed-exercised rats).
|
Effect of cigarette smoke and exercise on the abortion
As shown in Fig. 2 the exercise and smoke alone did not have significant effect on number of abortion. However, in rats in group which exposed to cigarette smoke and exercised simultaneously, the number of abortion was increased significantly (P<0.001).This finding indicated that the exercise in the smoke exposure had negative effect on the fetal growth.
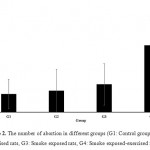 |
Figure 2: The number of abortion in different groups (G1: Control group, G2: Exercised rats, G3: Smoke exposed rats, G4: Smoke exposed-exercised rats).
|
Effect of cigarette smoke and exercise on the birthweight and offspring weight after 60 days
Effects of cigarette smoke on birthweight are mainlyas a results of fetal growth limitation. The mechanisms underlying the effects of cigarette on fetalgrowth are not well defined and are likely to be multifactorial.Fig. 3 represents the effect of cigarette smoke and exercise on the birthweight of offspring. Results indicated that simultaneous cigarette smoke exposure and exercise, during pregnancy, resulted in decrease in birthweight of offspring. These results were in accordance with earlier results which indicated that, exercised rats which were in exposure of smoke during pregnancy resulted in lower offspring birthweight. Accordingly, exercise or smoke exposure alone showed negative effect on birthweight of offspring, however were not significant.
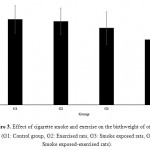 |
Figure 3: Effect of cigarette smoke and exercise on the birthweight of offspring (G1: Control group, G2: Exercised rats, G3: Smoke exposed rats, G4: Smoke exposed-exercised rats).
|
Fig. 4 also represents the effect of smoke and exercise alone and simultaneously on the offspring weight after 60 days. Like earlier results, exercise alone did not show significant effect on offspring weight after 60 days. However, surprisingly in the case of offspring weight after 60 days, exposure with smoke during pregnancy showed higher negative effect than rats which exercised in the exposure of cigarette smoke
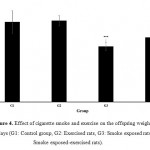 |
Figure 4: Effect of cigarette smoke and exercise on the offspring weight after 60 days (G1: Control group, G2: Exercised rats, G3: Smoke exposed rats, G4: Smoke exposed-exercised rats).
|
Effect of cigarette smoke and exercise on the placenta tissue
It is well recognized that smoking during pregnancy resulted in fetal hypoxia and increases the risk of perinatal problems.The placental pathologies relatedto maternal smoking areessential in many of the adverse developmental effects.Maternal smoking alters the placenta blood flow and balance between proliferation and differentiation of the cytotrophoblast. Furthermore, the changes in gene and protein expression in the cytotrophoblast, regulates cellular responses to oxygen tension, are also observed. These changes were seen in the placentas of smokers in less severe than women exposed to secondhand smoke during pregnancy[18-21].Previous studiesshowed that smokinginduces a generalized dysfunction of both villous andinvasive trophoblasts in early pregnancy. Biochemical markersof placental function show that maternal levels of oestriol,oestradiol, HCG, and humanplacental lactogen (HPL) hormones were lower in smokers thannon-smokers pregnant women [22,23]. Literature review showed that placental developmentis influenced by a change of environmental factors whichalter oxygen tension and placentalperfusion such as chronic pulmonary disease, diabetes,smoking, diet and sustained exercise. Furthermore, many reports indicated that maternal smoking has been associated with thickening of the trophoblastic basement membrane, increased collagen in the villous mesenchyme and decreased vascularization of the placenta[24-26].Placenta decidual thickness was measured in the all of four groups.Results indicated that, placenta decidual thickness in rats which were in the cigarette smoke exposure, exercised, or rats which exercised in the exposure of smoke did not change significantly in comparison with rats in control group.Results also showed that, in all of treated groups the number of blood vessels was decreased, however, only the number of blood vessels in rats exposed in cigarette smoke and exercised was decreased significantly (P<0.05).
Table 1: Effect of cigarette smoke and exercise on the placenta tissue
| Group | G1 | G2 | G3 | G4 |
| Placenta decidua thickness (µm) | 502.28 ± 148.8 | 489.5 ± 517 | 119.7 ± 517.9 | 456.5 ± 112.1 |
| Blood vessels number | 17.3 ±7.1 | 15.1 ± 3.9 | 11.1 ± 5.4 | 9.3 ± 4.6 |
(G1: Control group, G2: Exercised rats, G3: Smoke exposed rats, G4: Smoke exposed-exercised rats).
Conclusions
Cigarette smoke is a complex mixture of toxic chemicals including nicotine, carbonmonoxide, and several renowned carcinogens and mutagens which absorbed through the pulmonary vasculature and transported via the bloodstream. These toxins cause cytotoxicity, genotoxicity, and tumorigenicity throughout the body.Furthermore,in addition to the harmful effects of on cardiovascular and pulmonary physiology, cigarette smoking affects the reproductive system.Smoking have been found to be associated with infertility, ectopic pregnancy, and spontaneous abortion menstrual abnormalities.Results of the present study indicated that, exposure to cigarette smoke and exercise simultaneously showed synergic negative effect on placenta tissue, HCG hormone level, birthweight and weight of offspring weight after 60 days. Therefore, it is suggested that animals or humans which are in the exposure of cigarette smoke avoided from exercise.
Acknowledgments
The authors would like to thank the authorities of Physiology and pharmacology department, Faculty of Medicine,Zanjan University of Medical Sciences; Zanjan, Iran.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
References
- Hecht SS. Cigarette smoking and lung cancer: chemical mechanisms and approaches to prevention. The lancet oncology 2002;3(8):461-9.
CrossRef - Conde-Agudelo A, Althabe F, Belizán JM, Kafury-Goeta AC. Cigarette smoking during pregnancy and risk of preeclampsia: a systematic review. American journal of obstetrics and gynecology 1999;181(4):1026-35.
CrossRef - Hecht SS. Cigarette smoking: cancer risks, carcinogens, and mechanisms. Langenbeck’s Archives of Surgery 2006;391(6):603-13.
CrossRef - Thun MJ, Lally CA, Calle EE, Heath CW, Flannery JT, Flanders WD. Cigarette smoking and changes in the histopathology of lung cancer. Journal of the National Cancer Institute 1997;89(21):1580-6.
CrossRef - Ambrose JA, Barua RS. The pathophysiology of cigarette smoking and cardiovascular disease: an update. Journal of the American college of cardiology 2004;43(10):1731-7.
CrossRef - Health UDO, Services H. The health consequences of involuntary exposure to tobacco smoke: a report of the Surgeon General. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health 2006;709.
- Ockene IS, Miller NH. Cigarette smoking, cardiovascular disease, and stroke a statement for healthcare professionals from the American Heart Association. Circulation 1997;96(9):3243-7.
CrossRef - Sopori M. Effects of cigarette smoke on the immune system. Nature Reviews Immunology 2002;2(5):372-7.
CrossRef - Henningfield JE, Stapleton JM, Benowitz NL, Grayson RF, London ED. Higher levels of nicotine in arterial than in venous blood after cigarette smoking. Drug and alcohol dependence 1993;33(1):23-9.
CrossRef - Leonardi-Bee J, Britton J, Venn A. Secondhand smoke and adverse fetal outcomes in nonsmoking pregnant women: a meta-analysis. Pediatrics 2011;127(4):734-41.
CrossRef - Hanke W, Sobala W, Kalinka J. Environmental tobacco smoke exposure among pregnant women: impact on fetal biometry at 20–24 weeks of gestation and newborn child’s birth weight. International archives of occupational and environmental health 2004;77(1):47-52.
CrossRef - Roquer J, Figueras J, Botet F, Jimenez R. Influence on fetal growth of exposure to tobacco smoke during pregnancy. Acta Paediatrica 1995;84(2):118-21.
CrossRef - Kleinman JC, Pierre MB, Madans JH, Land GH, Schramm WF. The effects of maternal smoking on fetal and infant mortality. American Journal of Epidemiology 1988;127(2):274-82.
CrossRef - Clapp JF, Kim H, Burciu B, Lopez B. Beginning regular exercise in early pregnancy: effect on fetoplacental growth. American journal of obstetrics and gynecology 2000;183(6):1484-8.
CrossRef - Clapp JF, Kim H, Burciu B, Schmidt S, Petry K, Lopez B. Continuing regular exercise during pregnancy: effect of exercise volume on fetoplacental growth. American journal of obstetrics and gynecology 2002;186(1):142-7.
CrossRef - Jackson M, Gott P, Lye S, Ritchie JK, Clapp JR. The effects of maternal aerobic exercise on human placental development: placental volumetric composition and surface areas. Placenta 1995;16(2):179-91.
CrossRef - Bergmann A, Zygmunt M, Clapp J. Running throughout pregnancy: effect on placental villous vascular volume and cell proliferation. Placenta 2004;25(8):694-8.
CrossRef - Naeye RL. Effects of maternal cigarette smoking on the fetus and placenta. BJOG: An International Journal of Obstetrics & Gynaecology 1978;85(10):732-
CrossRef - Albuquerque CA, Smith KR, Johnson C, Chao R, Harding R. Influence of maternal tobacco smoking during pregnancy on uterine, umbilical and fetal cerebral artery blood flows. Early human development 2004;80(1):31-42.
CrossRef - Campbell S, Pearce J, Hackett G, Cohen-Overbeek T, Hernandez C. Qualitative assessment of uteroplacental blood flow: early screening test for high-risk pregnancies. Obstetrics & Gynecology 1986;68(5):649-53.
- Abel EL. Smoking during pregnancy: a review of effects on growth and development of offspring. Human Biology 1980:593-625.
- Michnovicz JJ, Hershcopf RJ, Naganuma H, Bradlow HL, Fishman J. Increased 2-hydroxylation of estradiol as a possible mechanism for the anti-estrogenic effect of cigarette smoking. New England Journal of Medicine 1986;315(21):1305-9.
CrossRef - Supervia A, Nogues X, Enjuanes A, Vila J, Mellibovsky L, Serrano S, et al. Effect of smoking and smoking cessation on bone mass, bone remodeling, vitamin D, PTH and sex hormones. Journal of Musculoskeletal and Neuronal Interactions 2006;6(3):234.
- Mochizuki M, Maruo T, Masuko K, Ohtsu T. Effects of smoking on fetoplacental-maternal system during pregnancy. American journal of obstetrics and gynecology 1984;149(4):413-20.
CrossRef - Genbacev O, Mcmaster MT, Zdravkovic T, Fisher SJ. Disruption of oxygen-regulated responses underlies pathological changes in the placentas of women who smoke or who are passively exposed to smoke during pregnancy. Reproductive Toxicology 2003;17(5):509-18.
CrossRef - Zdravkovic T, Genbacev O, Mcmaster M, Fisher S. The adverse effects of maternal smoking on the human placenta: a review. Placenta 2005;26:S81-S6.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





